As you well know, one of the most determining factors for the success of an endodontic treatment is the eradication of all microorganisms from the root canal system, that is, endodontic irrigation. This procedure is essential for cleaning the ducts and is performed by using isolated and combined chemical agents. In this article we will develop a detailed review of the most common endodontic irrigants that are applied to the root canal with their pros and cons, trying to find the chemical agent that best suits each of your cases... Shall we start?
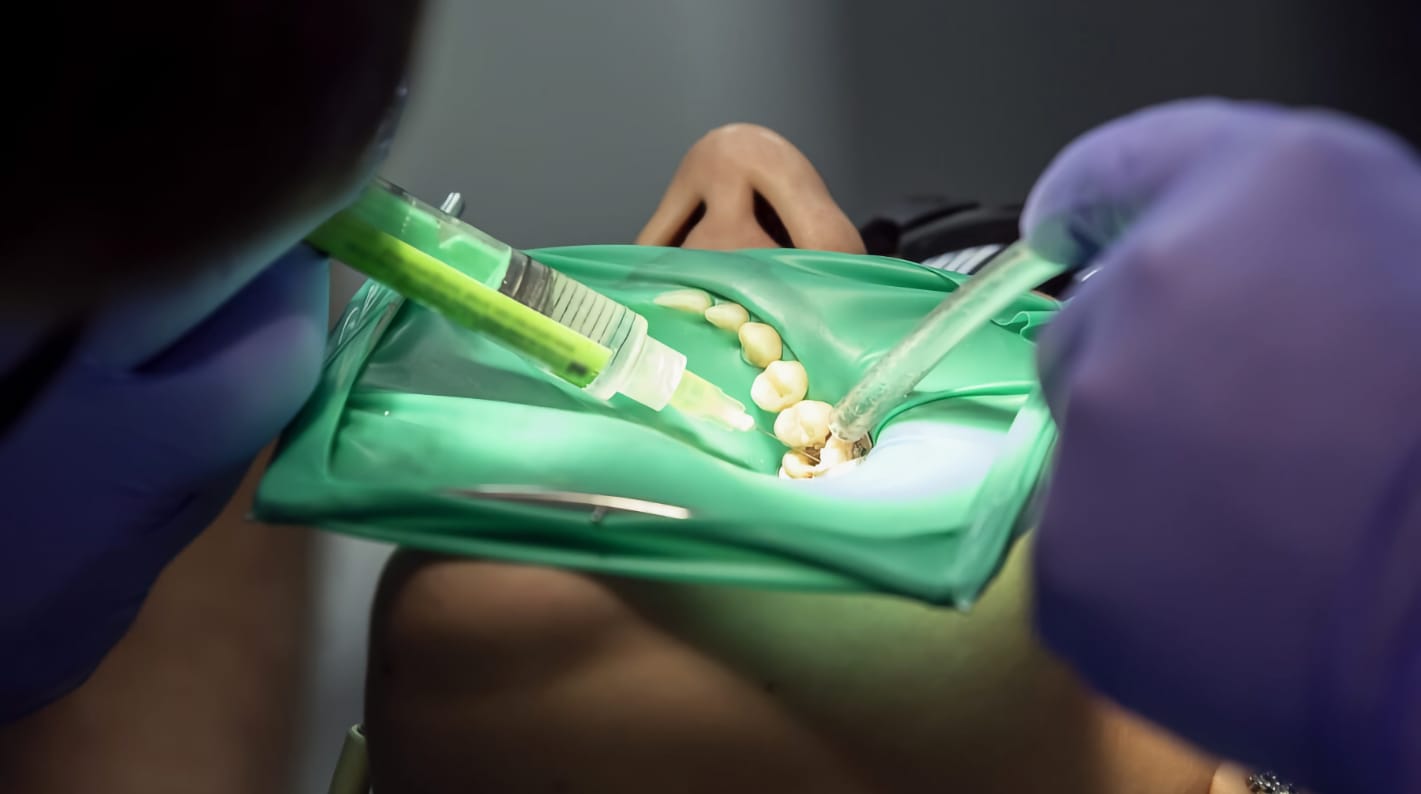
Characteristics that endodontic irrigating solutions must meet
- Low toxicity, should not be toxic to periradicular tissues.
- Bactericidal and bacteriostatic, also effective against fungi and spores.
- Low surface tension to penetrate the root canal system.
- Lubricant, should reduce friction during instrumentation.
- Rinsing action.
- Dissolution of organic material such as collagen from dentin, pulp tissue, and biofilm.
- Dissolution of inorganic tissue (dentin).
- Be easy to apply.
- The cost should not be too high.
The irrigation in endodontics is of great importance both in its technique and in the solutions it uses to remove vital or necrotic pulp remains , microorganisms and other remains of the instrumentation . Whether you perform manual or mechanized instrumentation, none by itself can clean the root canal, much less in critical areas such as the apical end of curved canals, for which reason instrumentation and irrigation are inseparably linked. Let's get into context!
The pulp and dentin are originally sterile thanks to the protection provided by a coating of enamel and cement. When this protection is lost in situations such as caries, fractures, fissures or does not exist naturally, the dentino-pulp complex is exposed to the oral environment where there are a large number of microorganisms that can contaminate it, These enter through the dentinal tubules, either due to the existence of periodontal disease, anachoresis, or direct pulp exposure.
Among the most direct routes of entry of microorganisms into the pulp space are dental caries and periodontal disease. If these microorganisms persist in the root canal, they will undoubtedly lead to endodontic treatment failure. In the same way, a poorly performed filling will accumulate inflammatory exudates that come from the periapical region, generating an environment conducive to the growth and proliferation of microorganisms.
That said, it is important to consider that any injury to the pulp can trigger an inflammatory response, microorganisms being the main etiological agent, even if the irritants are physical, thermal, or chemical. Now that we have recalled the basics, let's talk about root canal irrigation.
Efficacy of root canal irrigation
How to achieve effective endodontic irrigation?
- The needle/tip should be able to penetrate the canal to a certain depth. Take a look at these sterile Irrigation tips VDW Zipperer that can be very useful to you.
- The diameter of the needle/tip, both internal and external, should not be too large.
- Logically, it is easier to irrigate large-diameter canals than small-diameter canals.
- The pressure in the application of the irrigating solution influences, the narrower needles require more pressure and the irrigant reaches a higher speed, being more efficient.
- The irrigant should not be viscous.
Check out the best-selling chelating agents and endoactivators:
Irrigating solutions in endodontics
Among the irrigating solutions most used in endodontic treatments we can find the following:
- Sodium hypochlorite
- Chlorhexidine
- MTAD and Tetraclean
- Ethylenediaminetetraacetic acid (EDTA)
- Hydrogen peroxide
- Iodinated potassium iodide
Sodium hypochlorite (NaOCl)
What better than to start with a classic? Sodium hypochlorite originated in France around the year 1789 and was known as “eau de Javelle” in reference to the French city where it came from. In those years it was obtained in a very inefficient way, which is why it was necessary to devise other production methods. It was originally used as a wound irrigator during World War I and was later introduced to endodontics.
And why is it a classic? Simply because it is the most used irrigating solution due to its excellent antibacterial effect, its ability to dissolve necrotic tissue and vital pulp tissue, as well as the organic components of dentin and biofilms.
Sodium hypochlorite is usually used in variable concentrations ranging from 0.5 to 6%. Lower concentrations, for example 0.5 or 1%, are capable of dissolving necrotic tissue and higher concentrations increase this dissolution capacity, only that in addition to necrotic tissues, it also dissolves living tissue. And this is not always such a good idea!
Some authors recommend the use of sodium hypochlorite in conjunction with demineralizing substances, since by itself it provides minimal dentin removal. This demineralizer would help to remove the smear layer that forms after instrumentation from the surface of the root canal. Therefore, its joint use better cleans the most difficult-to-reach areas, including the dentinal tubules and lateral tubules.
There are very few cases in the literature of allergic reactions to sodium hypochlorite, after all, it is sodium and chlorine, which are elements that are always present in our physiology, however, in a very On rare occasions, hypersensitivities or contact dermatitis may occur. What do we do in this case? We do not use it and we do not use chlorhexidine either, but rather we looked for another irrigant with high antimicrobial efficacy as an alternative, such as iodized potassium iodide. Of course we should not use only water or alcohol since these are not effective, much less dissolve vital or necrotic tissues.
How can we make sodium hypochlorite even more effective in the root canal system? Although there are no clinical studies to support this fact yet, it is thought that increasing the temperature of a low concentration sodium hypochlorite solution could improve its ability to immediately dissolve tissue and remove organic debris from dentin more effectively. A study by the authors Sirtes G., Waltimo T., Schaetzle M., Zehnder M. corroborates that heated sodium hypochlorite improves its antimicrobial properties. But does heating hypochlorite increase systemic toxicity? At all, it is preferable to use a solution of sodium hypochlorite of low concentration heated to one of higher concentration.
Regarding the action time of hypochlorite, some authors point out that at a concentration of 5.25% and an exposure time of 5 minutes, it has the ability to remove biofilm. It is important to be aware of the fact that chlorine, being responsible for the antibacterial and dissolving capacity, is unstable and dissolves in the first two minutes, so it is important to continually replenish the irrigant.
Our recommendation:
Sodium Hypochlorite 5.25% Vidu
- 5.25% solution.
- Acting time from 3 to 5 minutes.
- Bottle contains 250 ml.
- Plastic footprint 0, the equivalent of the plastic of the bottle is collected from the Mediterranean Sea.

Chlorhexidine (CHX)
Chlorhexidine could also be considered a classic, although it developed much more recently than hypochlorite. It began to be used in the United Kingdom in 1953 as an antiseptic, disinfectant, treatment of skin infections , eyes and throat. It is a broad-spectrum antimicrobial that has proven its effectiveness against gram- and gram+ bacteria and is not only used as an endodontic irrigant, but is also used in dental therapy. periodontal, in implantology and cariology for the control of dental plaque, since its mechanism of action makes it adhere to the areas of the cell membrane with a negative charge and causes cell lysis.
Depending on the concentration in which chlorhexidine is used, its effects can be bacteriostatic or bactericidal.
Chlorhexidine in high concentration > Bactericidal effect
It has the power to damage the cell membrane by acting as a detergent and causing precipitation of the cytoplasm.
Low concentration chlorhexidine > Bacteriostatic effect
It causes the release of substances such as potassium and phosphorus that have low molecular weight, but does not irreversibly damage the cell. It is also capable of altering the metabolism of bacteria by preventing the transport of the sugar phosphotransferase system (PTS) and inhibiting acid production in the case of some bacteria.
Chlorhexidine also has a very interesting property which is antimicrobial substantivity, that is, it binds to dentin maintaining a sustained antimicrobial activity, therefore, it is used As an intracanal drug/irrigant it has the ability to delay coronal recontamination of the canal system. It is ideal especially in the case of endodontic retreatment.
Chlorhexidine used as an endodontic irrigant, both in liquid and gel, has different antibacterial properties depending on its concentration. Compared to hypochlorite, these properties do not have major differences, however, chlorhexidine is not capable of dissolving tissues and therefore sodium hypochlorite is still a better option.
Sometimes an irrigation protocol is suggested with which certain precautions must be taken due to the chemical interactions that may be generated:
- Irrigate with sodium hypochlorite to dissolve organic components.
- Irrigate with EDTA to remove smear layer.
- Irrigate with chlorhexidine to increase the antimicrobial spectrum of activity and add substantivity.
When mixing sodium hypochlorite with chlorhexidine an acid-base reaction is produced that forms a neutral insoluble precipitate that is thought to interfere with proper apical sealing, and a colour change is also produced. On the other hand, the mixture of chlorhexidine and EDTA forms a salt. Therefore, it is advisable to dry as well as possible with paper points before the final irrigation with chlorhexidine.
A precaution to consider is that chlorhexidine can cause an allergic reaction in approximately 2% of patients, although this data considers chlorhexidine used on the skin and not in a root canal.
We recommend:
Canalpro CHX 2% Chlorhexidine Coltene
- Great antibacterial efficacy against E. Faecalis bacteria and fungi.
- Practical dispenser system.
- Blue colour.
- Available in 100 and 500 ml bottles.

MTAD and Tetraclean
These irrigants are based on a mixture of antibiotics, citric acid, and detergent. MTAD, it is an irrigating solution that has the ability to remove smear layer and also disinfect the canal system. Tetraclean, similar to MTAD, which only differs in the concentration of the antibiotic doxycycline (150 mg/5ml in the case of MTAD and 50 mg/5ml in the case of Tetraclean) and in the type of detergent.
Ethylenediaminetetraacetic acid (EDTA)
In 1935, the Austrian chemist Ferdinand Munz synthesized for the first time ethylenediaminetetraacetic acid, fortunately better known as EDTA, which has the ability to chelate and remove the mineralized portion of the smear layer, which is why it is frequently used as an irrigating solution in endodontics. EDTA has multiple applications in various industries due to its ability to sequester metal ions, that is, it is a chelating agent.
The mechanism of action of EDTA is the extraction of proteins from the surface of bacteria by combining with the metal ions of their cell cover, causing their death.
EDTA is often used in combination with other endodontic irrigants, for example, sodium hypochlorite, since it alone is not capable of effectively removing organic components from smear layer. This irrigant is usually used in a 17% concentration and has the ability to remove smear layer when it is in direct contact with the root canal wall for less than 1 minute, it has the power to decalcify up to 50 μm, that is, it is self-limiting and this is sufficient for the opening of a fine occluded duct.
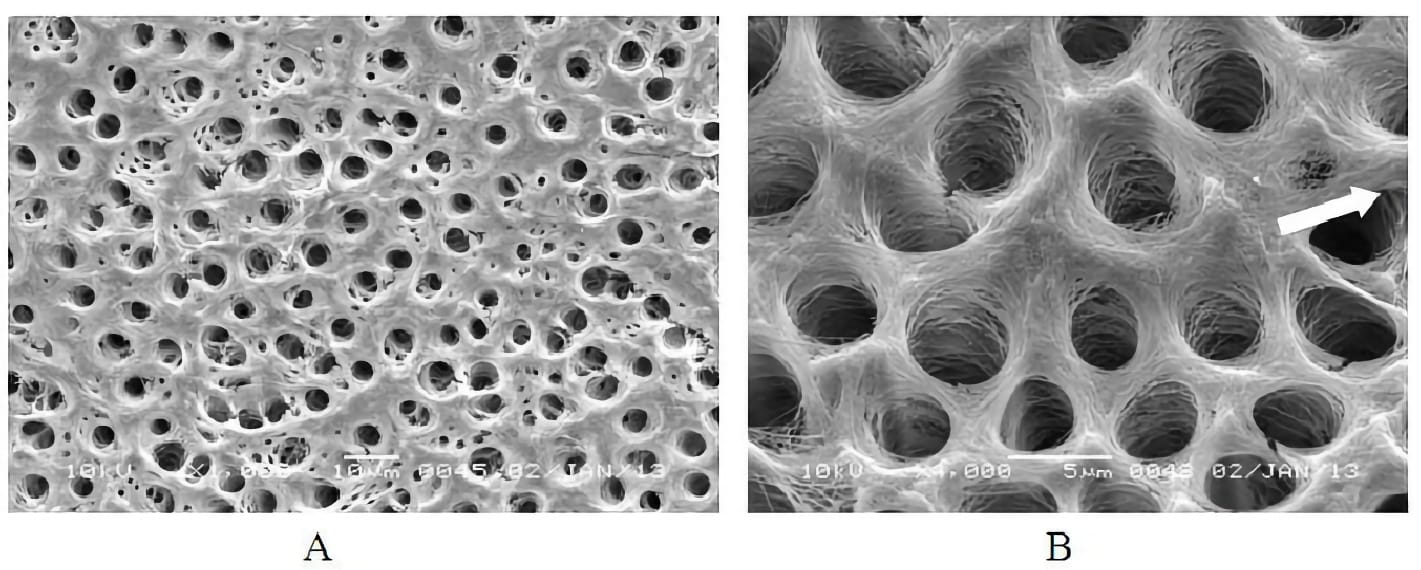
Image: The apical third is observed after irrigation with 17% EDTA and 2.5% NaOCL where a complete removal of the smear layer can be seen with patent dentinal tubules with cracks and destruction of the intertubular dentin. Image A x1000 and image B x4000.
Source: A Newly Prepared Solution for the Removal of the Smear Layer - Scientific Figure on ResearchGate.
Regarding the interactions that could occur with other irrigants such as sodium hypochlorite, it is concluded that both irrigants should be used separately, since EDTA causes sodium hypochlorite to lose its ability to dissolve tissues.
Recommended product:
EDTA 18% IndiSpense Ultradent
- It is a chelating conditioner that contains a moisturizing agent to the walls of the root canal.
- 18% peroxide-free solution.
- Efficiently removes smear layer.
- 30 ml syringe.

Hydrogen peroxide and iodized potassium iodide
In addition, there are other endodontic irrigants that are perhaps less common, such as hydrogen peroxide in concentrations of between 3 and 5% that act against bacteria, viruses and yeasts. On the other hand, iodinated potassium iodide (IKI) is used as a disinfectant in endodontics thanks to its great antibacterial properties and minimal toxicity. Both hydrogen peroxide and iodinated potassium iodide have some drawbacks that it is good to take into consideration when making the choice. The first, H2O2, when used with sodium hypochlorite, reacts by forming bubbles due to the oxygen that is released in the chemical reaction of the two liquids. The second, IKI, can cause severe allergic reactions and also stains the dentin.
In conclusion, and now that we know the common endodontic irrigants, you probably agree that sodium hypochlorite seems to be the ideal irrigating solution, since which has the exclusive capacity to dissolve the organic components of the smear layer and also the necrotic tissue. As if that were not enough, it is capable of destroying sessile endodontic pathogens in biofilms and in dentinal tubules in a way that is at least comparable to that of chlorhexidine. Its argument against it is undoubtedly cytotoxicity when it comes into contact with soft tissues and although the adverse effects it can cause are not very frequent, extrusion of hypochlorite to the periapex or accidental injection produce serious symptoms. that justify the rigid monitoring of a preventive protocol and an action protocol in case of accidents.
Finally, we cannot stop recommending what we could call an “endodontic gadget” that will help you safely enhance the irrigants you use in root canals.
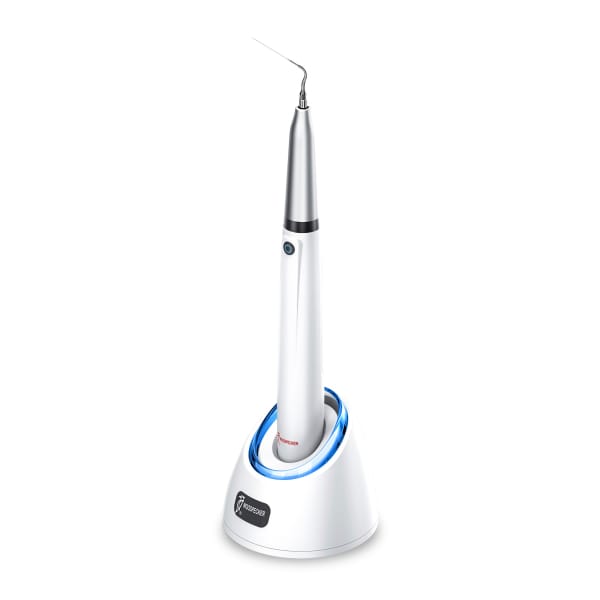
Endo 1 Woodpecker Ultrasonic Endoactivator
Woodpecker's Endo 1 Ultrasonic Endoactivator is an essential element for your endodontic treatments, since it allows the irrigants to penetrate deeply into the root canal system thanks to ultrasonic waves, making your irrigation even more efficient.
- Ergonomic, lightweight and wireless.
- The head can be disassembled and sterilized.
- Its ultrasonic vibration is high frequency, reaching 45 KHz and with a reduced wave amplitude of 25 µm.
- High capacity battery that allows 4 hours of continuous work and 6 months of standby.
- Compatible with Satelec system.
This is the end of our article today on the irrigant solutions frequently used in endodontics. Remember that at Dentaltix we have a wide variety of irrigants and irrigation needles that will help you achieve success in your endodontic treatments. If you want to always be informed about the latest news in the dental sector, we invite you to follow us on our social media and to contact us in case of any questions or comments. Until next time!